Miriam Santer
Eczema Care Online behavioural interventions to support self-care for children and young people: two independent, pragmatic, randomised controlled trials
Santer, Miriam; Muller, Ingrid; Becque, Taeko; Stuart, Beth; Hooper, Julie; Steele, Mary; Wilczynska, Sylvia; Sach, Tracey H.; Ridd, Matthew J.; Roberts, Amanda; Ahmed, Amina; Yardley, Lucy; Little, Paul; Greenwell, Kate; Sivyer, Katy; Nuttall, Jacqui; Griffiths, Gareth; Lawton, Sandra; Langan, Sinéad M.; Howells, Laura M.; Leighton, Paul; Williams, Hywel C.; Thomas, Kim S.
Authors
Ingrid Muller
Taeko Becque
Beth Stuart
Julie Hooper
Mary Steele
Sylvia Wilczynska
Tracey H. Sach
Matthew J. Ridd
AMANDA ROBERTS Amanda.Roberts@nottingham.ac.uk
Research Assistant
Amina Ahmed
Lucy Yardley
Paul Little
Kate Greenwell
Katy Sivyer
Jacqui Nuttall
Gareth Griffiths
Sandra Lawton
Sinéad M. Langan
LAURA HOWELLS LAURA.HOWELLS1@NOTTINGHAM.AC.UK
Senior Research Fellow
PAUL LEIGHTON PAUL.LEIGHTON@NOTTINGHAM.AC.UK
Associate Professor of Applied Health Services Research
HYWEL WILLIAMS HYWEL.WILLIAMS@NOTTINGHAM.AC.UK
Professor of Dermato-Epidemiology
Professor KIM THOMAS KIM.THOMAS@NOTTINGHAM.AC.UK
Professor of Applied Dermatology Research
Abstract
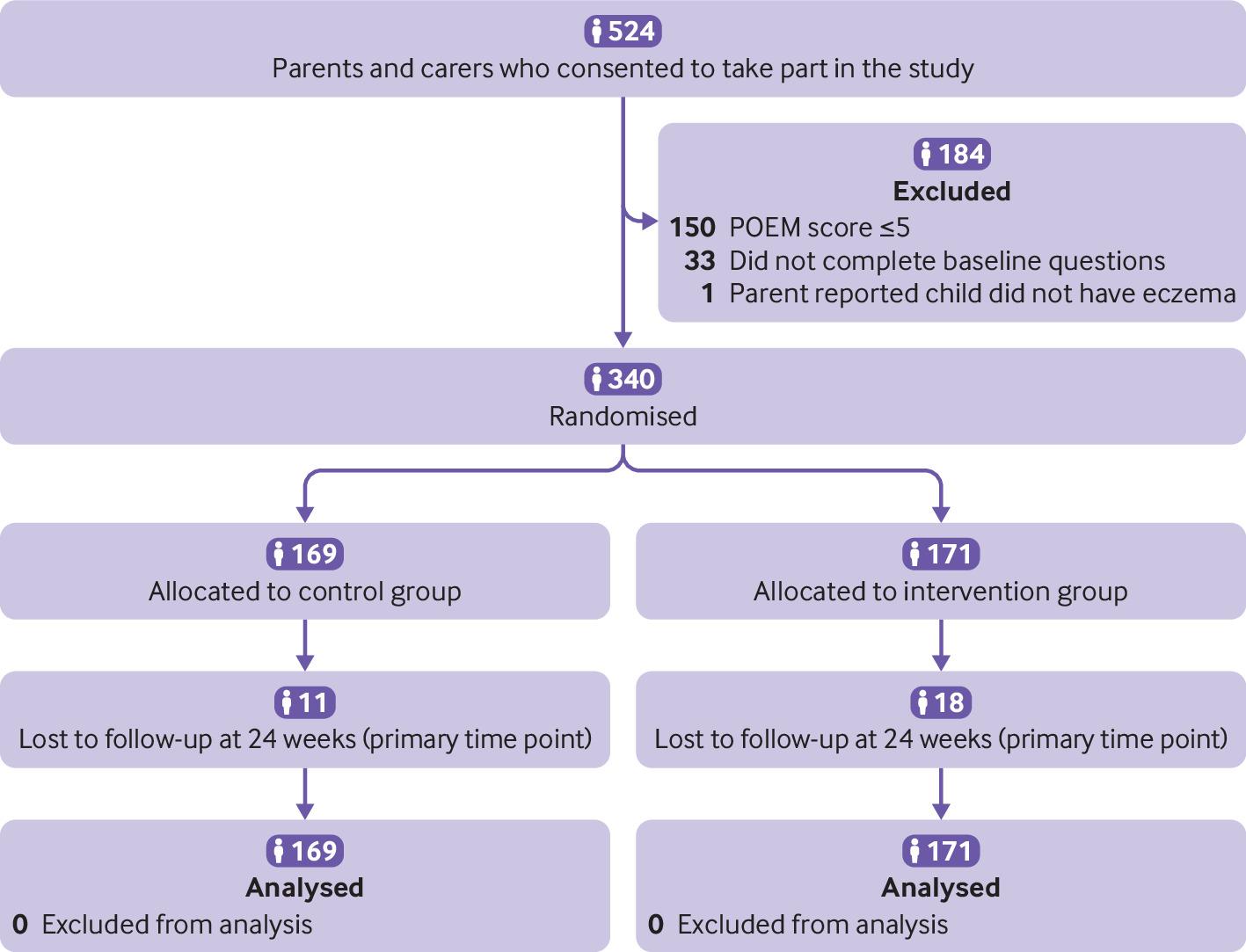
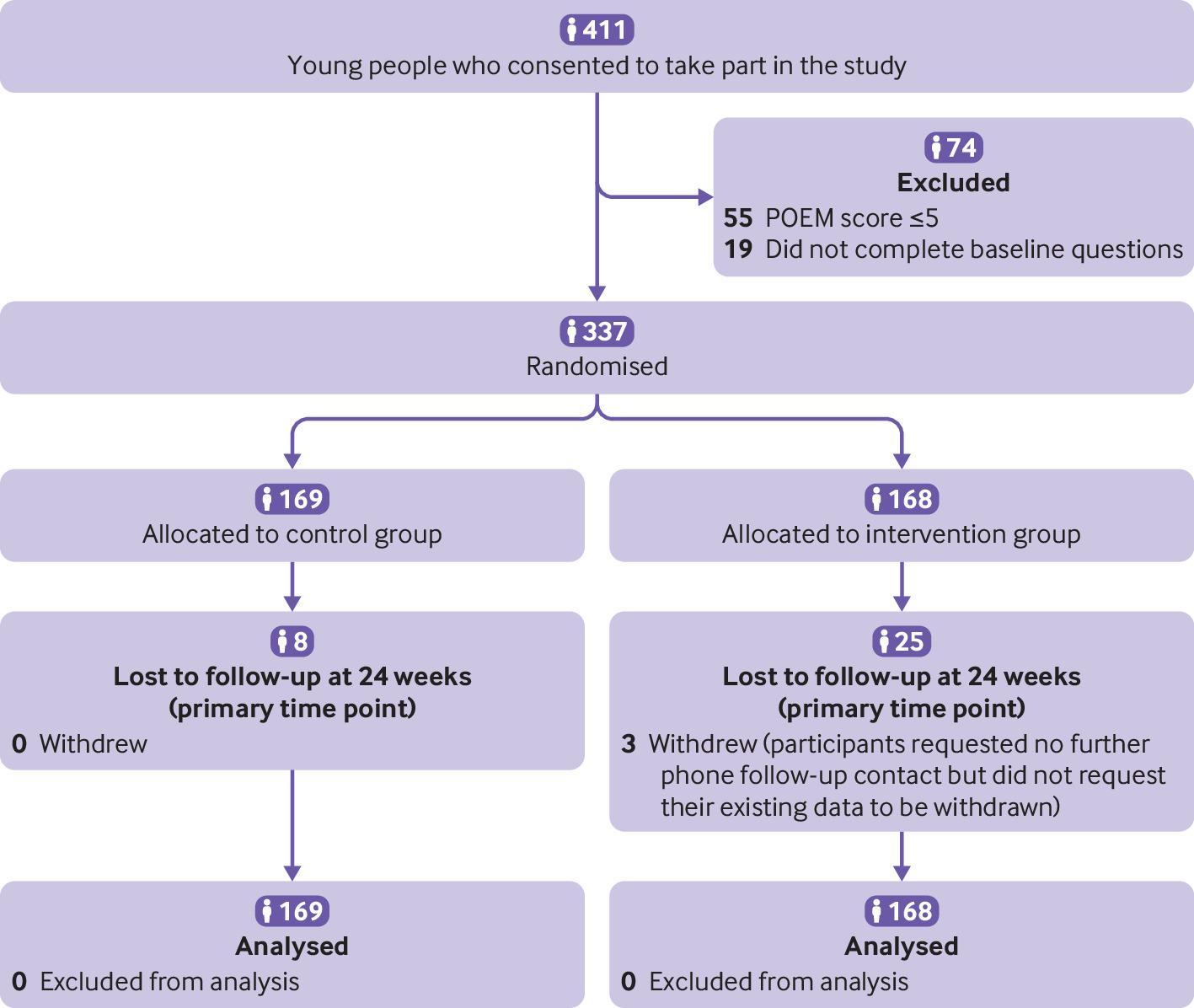
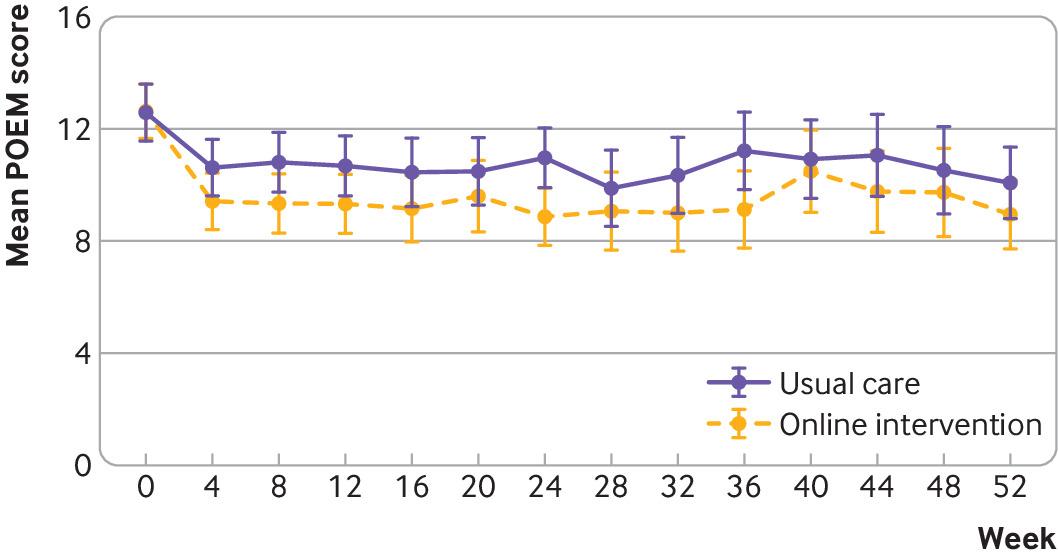
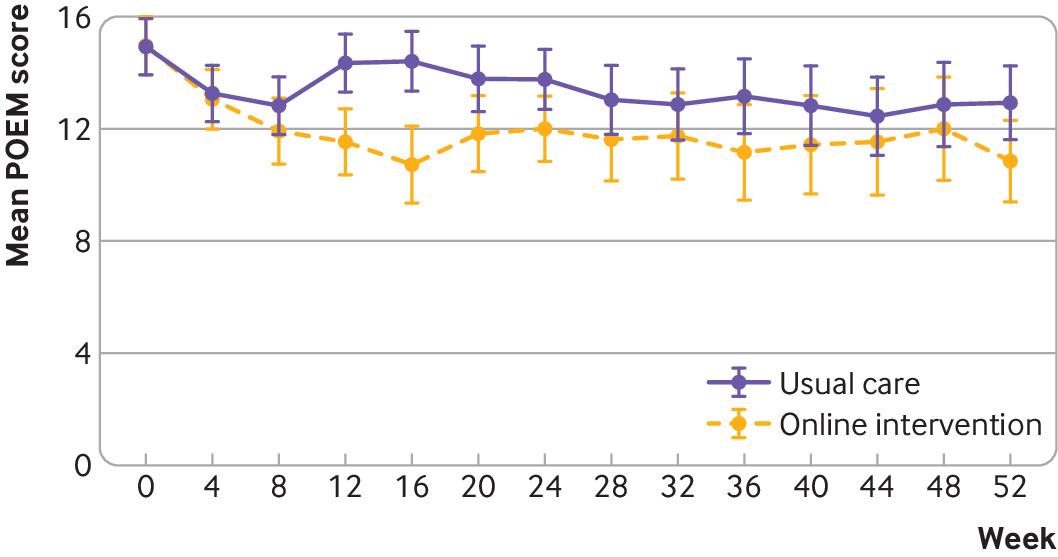
Objective: To determine the effectiveness of two online behavioural interventions, one for parents and carers and one for young people, to support eczema self-management. Design: Two independent, pragmatic, parallel group, unmasked, randomised controlled trials. Setting: 98 general practices in England. Participants: Parents and carers of children (0-12 years) with eczema (trial 1) and young people (13-25 years) with eczema (trial 2), excluding people with inactive or very mild eczema (≤5 on POEM, the Patient-Oriented Eczema Measure). Interventions: Participants were randomised (1:1) using online software to receive usual eczema care or an online (www.EczemaCareOnline.org.uk) behavioural intervention for eczema plus usual care. Main outcome measures: Primary outcome was eczema symptoms rated using POEM (range 0-28, with 28 being very severe) every four weeks over 24 weeks. Outcomes were reported by parents or carers for children and by self-report for young people. Secondary outcomes included POEM score every four weeks over 52 weeks, quality of life, eczema control, itch intensity (young people only), patient enablement, treatment use, perceived barriers to treatment use, and intervention use. Analyses were carried out separately for the two trials and according to intention-to-treat principles. Results: 340 parents or carers of children (169 usual care; 171 intervention) and 337 young people (169 usual care; 168 intervention) were randomised. The mean baseline POEM score was 12.8 (standard deviation 5.3) for parents and carers and 15.2 (5.4) for young people. Three young people withdrew from follow-up but did not withdraw their data. All randomised participants were included in the analyses. At 24 weeks, follow-up rates were 91.5% (311/340) for parents or carers and 90.2% (304/337) for young people. After controlling for baseline eczema severity and confounders, compared with usual care groups over 24 weeks, eczema severity improved in the intervention groups: mean difference in POEM score −1.5 (95% confidence interval −2.5 to −0.6; P=0.002) for parents or carers and −1.9 (−3.0 to −0.8; P<0.001) for young people. The number needed to treat to achieve a 2.5 difference in POEM score at 24 weeks was 6 in both trials. Improvements were sustained to 52 weeks in both trials. Enablement showed a statistically significant difference favouring the intervention group in both trials: adjusted mean difference at 24 weeks −0.7 (95% confidence interval −1.0 to −0.4) for parents or carers and −0.9 (−1.3 to −0.6) for young people. No harms were identified in either group. Conclusions: Two online interventions for self-management of eczema aimed at parents or carers of children with eczema and at young people with eczema provide a useful, sustained benefit in managing eczema severity in children and young people when offered in addition to usual eczema care. Trial registration: ISRCTN registry ISRCTN79282252.
Citation
Santer, M., Muller, I., Becque, T., Stuart, B., Hooper, J., Steele, M., …Thomas, K. S. (2022). Eczema Care Online behavioural interventions to support self-care for children and young people: two independent, pragmatic, randomised controlled trials. BMJ, 379, Article e072007. https://doi.org/10.1136/bmj-2022-072007
| Journal Article Type | Article |
|---|---|
| Acceptance Date | Oct 31, 2022 |
| Online Publication Date | Dec 7, 2022 |
| Publication Date | Dec 8, 2022 |
| Deposit Date | Jan 17, 2023 |
| Publicly Available Date | Jan 17, 2023 |
| Journal | BMJ |
| Electronic ISSN | 0959-8138 |
| Peer Reviewed | Peer Reviewed |
| Volume | 379 |
| Article Number | e072007 |
| DOI | https://doi.org/10.1136/bmj-2022-072007 |
| Public URL | https://nottingham-repository.worktribe.com/output/14879686 |
| Publisher URL | https://www.bmj.com/content/379/bmj-2022-072007 |
Files
Eczema Care Online
(226 Kb)
Image
Eczema Care Online
(244 Kb)
Image
Eczema Care Online
(170 Kb)
Image
Eczema Care Online
(171 Kb)
Image
Eczema Care Online
(747 Kb)
PDF
Publisher Licence URL
https://creativecommons.org/licenses/by/4.0/
You might also like
The long-term safety of topical corticosteroids in atopic dermatitis: A systematic review
(2023)
Journal Article
Public views are needed for skin colour scales
(2023)
Journal Article
Downloadable Citations
About Repository@Nottingham
Administrator e-mail: digital-library-support@nottingham.ac.uk
This application uses the following open-source libraries:
SheetJS Community Edition
Apache License Version 2.0 (http://www.apache.org/licenses/)
PDF.js
Apache License Version 2.0 (http://www.apache.org/licenses/)
Font Awesome
SIL OFL 1.1 (http://scripts.sil.org/OFL)
MIT License (http://opensource.org/licenses/mit-license.html)
CC BY 3.0 ( http://creativecommons.org/licenses/by/3.0/)
Powered by Worktribe © 2024
Advanced Search